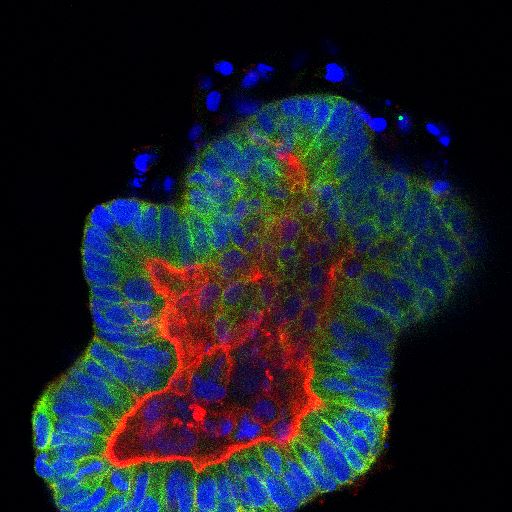
The POPYNA project aims to understand intra-tumor phenotypic heterogeneity in colorectal cancer. Currently funded by the prestigious Marie Skłodowska-Curie Actions (MSCA) fellowship, our goal is to investigate POPulation dYNAmics in colorectal cancer to uncover key players in tumor development, treatment response, and resistance.
It’s well-established that for a malignant tumor (cancer) to grow, it must accumulate DNA mutations enabling tumor cells to proliferate uncontrollably, evade cell death, and eventually metastasize. However, DNA mutations are not the sole drivers of these capabilities. Epigenetic alterations, transcriptional regulation, protein modifications, and other mechanisms also contribute to the acquisition of malignant traits by tumor cells.
Today, we understand that within a malignant tumor, subsets of cells (subpopulations) exhibit distinct phenotypic characteristics not associated to DNA mutations. This means that not all cells within a tumor are identical, suggesting that different subpopulations may play varying roles in tumor development and dissemination. Moreover, these subpopulations may respond differently to specific treatments. Notably, these subpopulations exhibit plasticity, allowing them to adjust their phenotypic profiles in response to environmental cues, such as treatment.
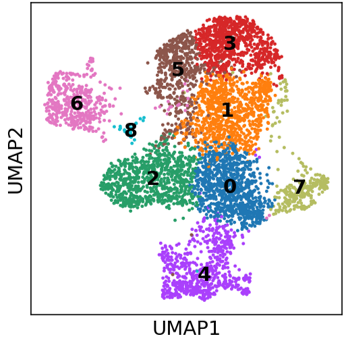
During this project, we will utilize colorectal cancer organoids as in vitro models to identify subpopulations present during tumor development and treatment. Additionally, we aim to elucidate the interactions between subpopulations present in the same tumor and their hierarchical relationships. To achieve this, we will employ 3D cell cultures (organoids or tumoroids) as well as state-of-the-art technologies such as single-cell RNA sequencing, CRISPR editing, and live confocal imaging.
We hope that the findings from this project will yield new insights into intrinsic features of tumor growth and provide evidence supporting the development of novel treatment strategies targeting tumor subpopulations rather than specific proteins.

